Resumen
El cáncer de pulmón es la causa principal de mortalidad por cáncer en los países occidentales. Aproximadamente el 80% de los cánceres de pulmón son los llamados no microcíticos (CPCNP). La cura es poco probable en pacientes con CPNM con estadio localmente avanzado y que no son candidatos quirúrgicos, con una tasa de supervivencia de 3 años del 27% en aquellos pacientes que reciben quimioterapia y radioterapia concomitante. En el momento del diagnóstico, al menos el 40% de los pacientes se diagnostican en una etapa avanzada y una tercera parte en enfermedad localmente avanzada (estadio III). Solo el 25-30% del NSCLC son candidatos para cirugía de intención curativa. Los resultados del estadio IIIA con tratamiento de inducción de la práctica clínica fuera del ensayo clínico muestran una supervivencia media de 22 meses y una tasa de supervivencia de 3 años del 34%. La resección quirúrgica completa, la bajada de estadio y la respuesta patológica completa son predictores de supervivencia a largo plazo después de la terapia neoadyuvante. La respuesta patológica completa después de la quimioterapia de inducción generalmente varía de 0% a 9.5%. En el estudio NADIM, un estudio multicéntrico abierto de fase II, de un solo brazo, destinado a evaluar la viabilidad, seguridad y eficacia de la quimioterapia neoadyuvante combinada y la inmunoterapia, obtenemos las tasas de pCR observadas sin precedentes (alrededor del 70%) y altamente prometedoras en el contexto de la terapia neoadyuvante del NSCLC con una bajada de estadio de alrededor del 90%. Creemos que es una de las modalidades de tratamiento más prometedoras en el estadio III del cáncer de pulmón que ha surgido en los últimos años es la quimioterapia combinada con inmunoterapia.
Abstract
Lung cancer is the primary cause of cancer mortality in western countries. Approximately 80% of lung cancers are non-small-cell lung cancer (NSCLC). The cure is unlikely in patients with NSCLC and locally advanced stage who are not surgical candidates, with a 3-year survival rate of 27% in those patients receiving chemotherapy and concomitant radiotherapy. At diagnosis, at least 40% of patients are diagnosed at an advanced stage and a third locally advanced disease (stage III). Only 25-30% of the NSCLC are candidates for curative-intent surgery. The results of stage IIIA with induction treatment of clinical practice outside the clinical trial show a median survival of 22 months and a 3-year survival rate of 34%. Complete surgical resection, tumor downstaging and pathologic complete response are predictors of long-term survival following neoadjuvant therapy. Pathologic complete response after induction chemotherapy generally ranges from 0% to 9.5%. In NADIM Study, a Phase II, single-arm, open-label multicenter study aimed to assess the feasibility, safety and efficacy of combined neoadjuvant chemotherapy and immunotherapy, we obtain pCR rates observed are unprecedented (around 70%) and highly promising in the context of neoadjuvant therapy of NSCLC with down-staging around 90%. We believe that one of the most promising treatment modalities in stage III in lung cancer that has emerged in recent years is chemo-immunotherapy.
Palabras clave: Estadio localmente avanzado; Cáncer de pulmón; Inmunoterapia.
Keywords: Locally advanced stage; Lungcancer; Inmunotherapy.
INTRODUCTION
Lung cancer is the primary cause of cancer mortality in western countries. Approximately 80% of lung cancers are non-small-cell lung cancer (NSCLC).
In Spain occur about 18,800 new cases per year (1) and has been responsible for 19,513 deaths in 2006, twice the mortality of colon cancer (the most common tumor in absolute terms in Spain) (2) and NSCLC accounts for 85% of newly diagnosed cases. Most patients are diagnosed with unresectable disease and around 40% advanced (3) disease. The cure is unlikely in patients with NSCLC and locally advanced stage who are not surgical candidates, with a 3-year survival rate of 27% in those patients receiving chemotherapy and concomitant radiotherapy (4). On the contrary, in localized stages (stage I, II, IIIA) with surgical resection and cytostatic therapy, a survival of 5 years of 51% (5) is achieved and those with an absolute benefit in survival at 5 years of 5.4%, especially in patients with good performance status (PS) (6).
BACKGROUND NEOADJUVANT TREATMENTS
At diagnosis, at least 40% of patients are diagnosed at an advanced stage and a third locally advanced disease (stage III). We understand as locally advanced disease when the tumor exceeds the lung structures, but without clinical evidence of distant spread, and are a very heterogeneous group of patients with a controversial treatment based on a combination of surgery, chemotherapy and radiotherapy.
In the past, radiation therapy was considered standard therapy for patients with stage IIIA and IIIB but presented poor survival with poor local control and early development of distant disease. Patients with inoperable stage III treated with chest radiation therapy alone, had a median survival of 11.9 months, survival at 2 years of 10-20% and 3 years 5-10% (7).
Currently, there is no consensus on the best standard treatment and it has been demonstrated that the experience of the therapeutic team plays an important role in the decisions to take.
Only 25-30% of the NSCLC are candidates for curative-intent surgery. The rest are advanced local tumors or widespread metastases. Survival at 5 years depends, among other factors, on the size of the tumor and lymph node involvement. But even without mediastinal involvement, less than half of the patients survive more than 5 years and the majority dies of disseminated metastases.
Patients with stage IIIA disease with clinically evident N2 nodal spread have an overall 5-year survival rate of only 10%-15%, although this fall to 2%-5% in those with bulky mediastinal N2 involvement. The surgical management of stage IIIA NSCLC remains highly controversial and most patients with stage IIIB disease are generally considered inoperable. The aims of therapy in stage III NSCLC are to increase both locoregional and systemic control of the disease. As a matter of fact, it is reported that at least 80% of patients treated with local modalities alone will have micrometastases and will relapse. These aims could in some way be in conflict and may require different combined modality therapy sequencing strategies. Success in achieving them is measured in time of progression, survival and cure rate. Strategies that have been investigated include induction chemotherapy, concomitant chemoradiotherapy, intensified radiotherapy and adjuvant treatment. Since distant metastases remain the major site of failure, it is likely that more effective cytotoxic or other anti-tumor agents will be required further to improve current levels of response and survival (8). Meta-analysis has suggested that cisplatin-based induction chemotherapy prior to surgery reduces risk of death by 13 % and increased absolute 5-year survival rates by 5% (9). Neoadjuvant therapy has theoretical advantages: in vivo assessment of response to chemotherapy helps identify patients who will potentially benefit from adjuvant chemotherapy, early treatment of micrometastatic disease, reduction in drug resistance by early exposure to treatment and downstaging with improved resectability. Potential disadvantages include: delay in local therapy secondary to toxicity, risk progression in chemoresistant patients and pre-operative complications.
Several newly available chemotherapeutic agents are both highly active against NSCLC and potent radiosensitizers.
The results of stage IIIA with induction treatment of clinical practice outside the clinical trial show a median survival of 22 months and a 3-year survival rate of 34% (10). An EORTC study (11) with carboplatin and paclitaxel used as induction regimen in patients with biopsy-proven stage N2 non-small cell lung cancer of the 52 eligible patients, 33 patients responded, one CR and 32 PR, for an overall response rate of 64% (95% CI, 48%- 76%). In addition, there were 10 patients with no changes (10%) and 9 with progressive disease (17%). The median duration of survival was 20.5 months (95% CI, 16.1-31.2 months) with an estimated 1-year survival rate of 68.5% (95% CI, 55.2-81.7). Furthermore, phase II neoadjuvant studies of docetaxel alone, in combination with cisplatin or carboplatin, or in combination with platinum and gemcitabine have produced promising results, with more recently reported RRs ranging from 44 to 82% and rates of complete resection raging from 67 to 79% (12)
PROGNOSTIC FACTORS AFTER NEOADJUVANT THERAPY
Complete surgical resection (13,14), tumor downstaging and pathologic complete response are predictors of long-term survival following neoadjuvant therapy.
Pathologic complete response after induction chemotherapy generally ranges from 0% to 9.5%. Others higher complete response: one Martini (15) with 16.7% and one Kumar with 15% (16) are rare.
Andre analyzed a cohort of 702 patients with resected N2 disease and identified four negative factors: preoperative clinical N2 status, involvement of multiple lymph node levels, pathological T3 or T4 disease, and absence of preoperative chemotherapy (17). Choi et al (18), reviewed cases of pathologic proven N2 disease, complete resection rate was 83,2% and overall 5-year was 23,3%. Five-year recurrence –free survival was 19,6%. Among 19 clinicopathological prognostic factors, incomplete resection and non-downstaging after neoadjuvant therapy were unfavorable prognostic factors in univariate analyses. Clinical N2 status, multiple N2 nodes, and cell type of adenocarcinoma showed poor prognosis but were not statistically significant. Postoperative chemotherapy showed good prognosis but was not statistically significant. Multivariate analysis showed that significant favorable prognostic factors were complete resection and adjuvant chemotherapy (19). Experience of Memorial Sloan-Kettering Cancer Center confirms survival is significantly influenced by patient age, the median survival for complete resection 27.8 months compared with 11.4 months for incomplete resection, pathologic stage with 3-year survival for N0/N1 was 43.3% and 25.5% for N2 patients (20).
IMMUNOTHERAPY IN TREATMENT OF CANCER
An emerging hallmark of cancer is immunoevasion—the cancer cell’s ability to avoid destruction by the immune system. The three general categories of immunoevasive mechanisms include: (A) an insufficient number of T cells generated within the lymphoid compartment; (B) an insufficient number of T cells extravasating into the tumor; and (C) inhibition of T cells in the tumor microenvironment. The tumor microenvironment, in turn, offers three main immunoevasive tools: (1) surface membrane proteins that function as immune checkpoints, including PD-1, CTLA-4, lymphocyte-activation gene 3 (LAG-3) protein, T-cell immunoglobulin and mucin domain–containing protein 3 (TIM-3), B- and T-lymphocyte attenuator (BTLA), and the adenosine A2a receptor (A2aR); (2) the relationship between selected soluble factors and metabolic alterations, such as IL-10, transforming growth factor beta, adenosine, indoleamine 2,3-dioxygenase (IDO), and arginase; and (3) inhibitory cells, including cancer-associated fibroblasts (CAFs), regulatory T cells, myeloid-derived suppressor cells (MDSCs), and tumor-associated macrophages.
The immune response and the use of strategies to upregulate surface proteins, including programmed death 1 (PD-1), is a new approach for the treatment of tumors. PD-L1 overexpression has been observed in 40% – 50% of all NSCLC tumours, on the set of all stages and histologies (21). Targeted therapy to PD-1 receptor and to PD-L1 ligands is intended to inhibit their intervention and is an attractive therapeutic option in the locally advanced NSCLC stage, which can reactivate the host immune responses and allow good long-term control of the tumor (22).
In lung cancer, inhibition of the Check Point PD-1 pathway with antibodies directed against PD-1 or against its ligand, PD-L1, has showed preliminary and encouraged results that suggest a “class effect” and validate this pathway as a therapeutic target in NSCLC.
Results from cohorts of heavily pretreated NSCLC patients in phase I studies showed objective responses dose dependent, ranging from 10% to 32% (23).
Inhibition of the Checkpoint PD-1 pathway with antibodies against PD-1 or PD-L1 produces long lasting tumor response and stable disease as well, for more than 6 months.
Exploratory analysis of PD-L1 tumor expression and treatment response have confirmed the prevalence of > 40% of PD-L1 expression in NSCLC. Some studies suggest an association between treatment response and PD-L1 tumor expression before treatment.
However, PD-L1 expression role as a biomarker for response has not yet been validated. Immunotherapy with antibodies anti-PD-1 and anti-PD-L1 in many different tumors types has been, in general, well tolerated. Frequent adverse events related to the drug are limited episodes of grade 1 or 2 fatigue, diarrhea, rash, pruritus, nausea and decreased appetite.
In clinical trials, grade 3 or 4 adverse events related to the treatment occur in < 15% of patients. Inmune related adverse events treatment related are infrequent (<2%) and include pneumonitis, vitiligo, colitis, hepatitis, thyroiditis and hypophysitis (24)
Numerous ongoing trials are evaluating the combination of chemotherapy and checkpoint blockade in solid tumors, including melanoma, NSCLC, and SCLC. In untreated metastatic melanoma, a phase III study showed that ipilimumab (at 10 mg/kg) plus dacarbazine improved OS compared with dacarbazine alone (11.2 vs 9.1 months, respectively), but this was at the expense of higher toxicity and there was no ipilimumab-alone comparator arm. A phase II study showed that phased but not simultaneous ipilimumab plus platinum doublet chemotherapy (carboplatin/paclitaxel) improved immune-related PFS in patients with stage IIIB or IV NSCLC and extensive-stage SCLC, when compared with chemotherapy alone (25, 26). The choice of chemotherapy and dosing schedule are thus critical to optimizing outcomes of checkpoint blockade and chemotherapy combinations. With this in mind, a phase I four-cohort study evaluated first-line nivolumab at 10 mg/kg (N10) vs 5 mg/kg (N5) in combination with gemcitabine/cisplatin (N10) in advanced squamous-cell NSCLC, pemetrexed/cisplatin (N10) in advanced nonsquamous NSCLC, and paclitaxel/carboplatin (N5 vs N10) in combined cohorts of squamous and nonsquamous NSCLC (27). The toxicity profile was additive, representing effects of both nivolumab and chemotherapy. The ORR, PFS, and 1-year OS outcomes were acceptable. In particular the 1-year OS rate was 85% for the N5 paclitaxel/carboplatin group and 87% for the N10 pemetrexed/cisplatin group, which may reflect a positive signal.
A phase Ib study enrolled untreated patients with locally advanced or metastatic NSCLC to three treatment arms of atezolizumab plus chemotherapy, including carboplatin/pemetrexed, carboplatin/paclitaxel, and carboplatin/nab-paclitaxel (28). Atezolizumab at 15 mg/kg every 3 weeks was administered with standard chemotherapy for 4 to 6 cycles followed by atezolizumab maintenance or atezolizumab/pemetrexed maintenance in the carboplatin/pemetrexed arm. A preliminary analysis on 41 patients showed that the ORR was 64% (95% CI, 46.9–77.9) by RECIST, with the carboplatin/pemetrexed arm having the highest response rate at 75% (95% CI, 45–93). The four complete responses occurred in the carboplatin/nab-paclitaxel arm. The toxicity profile was as expected for chemotherapy, and no pneumonitis was observed. There was one grade 5 adverse event in a patient in the carboplatin/nab-paclitaxel arm who developed candidemia after prolonged neutropenia. Overall, the combination therapy response rates exceeded the 30% traditionally expected with platinum doublet chemotherapy; more mature data are forthcoming.
Several studies in patients with NSCLC suggested an association of increased immune cell infiltration into tumours with improved survival. In recent years, improved identification of antigenic targets, the addition of immunoadjuvants, and the production of more efficient delivery systems have resulted in more efficient vaccines, able to elicit a potent immune response, leading to the development of immunotherapy for the treatment of NSCLC (29, 30).
The adaptive immune response requires two signals between the antigen-presenting cells (APCs) and the effector T-cells. The first signal is mediated by the T-cell receptor and the major histocompatibility complex classes I or II antigenic peptide. The second signal is a co-stimulatory signal mediated by CD28 on the T-cell surface through binding of the B7 family members on APCs. Both signals result in the activation and clonal proliferation of T-cells.
In order to avoid autoimmunity, T-cell proliferation is tightly regulated. The balance between co-stimulatory signals mediated by CD28 and co-inhibitory signals via so called immune checkpoint receptors is crucial for the maintenance of self-tolerance and to protect tissues from damage during normal immune response. After activation, T-cells express cytotoxic T-lymphocyte antigen-4 (CTLA-4) and programmed cell death protein 1 (PD-1, cluster of differentiation 279 [CD279]), both so called immune checkpoint receptors.
CTLA-4 binds members of the B7 family with a much higher affinity than CD28 and down-regulates the T-cell response. It has been shown in pre-clinical models that one reason for the poor immunogenicity of many tumours such as lung cancer is CTLA-4 activity and that in vivo administration of antibodies to CTLA-4 can enhance antitumour immunity (31).
CD4+CD25+ regulatory T-cells (Treg) that express FOXP3 represent a group of T lymphocytes that is essential for maintaining self-tolerance (32). The transcription factor FOXP3 represses IL2 transcription and up-regulates expression of CTLA-4. FOXP3+CD25+CD4+ Treg cells constitutively express cell surface CTLA-4. CTLA4 thus maintains the immune system homeostasis by functioning as a major feedback inhibitor of T‑cell activation.
PD-1 is another immune checkpoint receptor expressed on activated T‑cells. Its physiological role is to dampen the immune response in order to protect against excessive inflammation and development of autoimmunity. PD‑1 is expressed in response to inflammation and is found in many tumours. Compared with CTLA-4, PD-1 modulates a later stage of the immune response. Instead of affecting the initial stage of T-cell activation (priming) in the regional lymph node, PD-1 regulates the activation of T‑cells in peripheral tissues. Like CTLA-4, PD-1 can be found on Treg lymphocytes and also on B lymphocytes and natural killer cells. PD-1 binds to its ligands PD-L1 (B7-H1) and PD-L2 (B7-DC), which are expressed on antigen presenting cells but more importanly, also on cancer cells.
While CTLA-4- and PD-1 expressing Tregs may play a critical role in maintaining self-tolerance, they also play a role in non-responsiveness to tumour antigens.
It is a recognised feature of cancer cells to escape immune surveillance by expressing ligands binding to immune checkpoint receptors and the development of therapies to enhance immunogenic activity towards tumours is a rational treatment strategy. The goal of checkpoint inhibitor therapies is not to activate the immune system to attack particular targets on tumour cells, but rather to remove inhibitory pathways that block effective antitumour T‑cell responses. Tregs have been shown to be present in tumours and coexist with primed effector T‑cells. Blockade of Tregs function via anti-CTLA-4 and anti-PD-1 has the potential to remove Tregs suppression and enhance antitumour immunogenicity (33, 34).
IMMUNE CHECKPOINT INHIBITOR AND CHEMOTHERAPY
Nivolumab (BMS-936558; anti-PD-1) is a fully human monoclonal immunoglobulin G4 (IgG4) antibody (HuMAb) that targets the cell surface membrane receptor PD-1. The co-inhibitory receptor PD-1, a member of the CD28 superfamily of molecules, has important T‑cell regulatory functions. It is inducibly expressed on activated T‑cells, B‑cells, a subset of myeloid cells and a fraction of T‑memory cells, and it has been shown to mediate inhibition of T‑cell responses in peripheral tissues and tumours. Engagement of PD-1 by its natural ligands, PD-L1 and PD-L2, results in an inhibition of T‑cell proliferation, survival and cytokine secretion (35, 36). Nivolumab abrogates this interaction between PD-1 and its ligands.
Nivolumab monotherapy has been approved for the treatment of advanced melanoma (FDA, EMA, and Japan) and previously treated squamous NSCLC (FDA, positive CHMP opinion). Nivolumab and ipilimumab improved PFS compared to nivolumab or ipilimumab alone in a study in melanoma (CA209067).
A phase I trial tested nivolumab in 296 patients with advanced solid cancers, including 129 NSCLC patients (37, 38). Nivolumab was administered intravenously once every 2 weeks at doses of 1, 3 or 10 mg/kg. Patients continued treatment for up to 96 weeks (12 cycles) or until unacceptable toxicity, confirmed complete response, confirmed disease progression, or withdrawal of consent. In the absence of clinical deterioration, patients could continue treatment after initial disease progression to allow for patterns of response consistent with immune-related response criteria. In the NSCLC cohort, with a long term median follow-up of 27.5 months (range, 21 to 54 months), median overall survival (OS) across nivolumab doses was 9.9 months. One- and 2-year OS rates were 42% and 24%, respectively, across doses and 56% and 45%, respectively, at the 3 mg/kg dose (n=37) being used for further clinical development. Among 22 (17%) patients with objective responses, estimated median response duration was 17.0 months. Response rates were similar in squamous and non-squamous NSCLC and in patients who received 3 or more prior therapies. Sixteen responding patients discontinued nivolumab for reasons other than progressive disease and 6 (38%) had responses lasting >30 weeks after their last dose. Grade 3-4 treatment-related adverse events occurred in 14% of patients. Three treatment-related deaths (2% of patients) occurred, each associated with pneumonitis (37).
Recently, the first randomized trials using nivolumab in comparison to standard of care docetaxel in the second line setting have been reported (39, 40).
The first one (checkmate 017) was focusing on squamous histology advanced NSCLC patients. 272 patients were assigned to receive nivolumab at a dose of 3 mg per kilogram of body weight every 2 weeks, or docetaxel at a dose of 75 mg per square meter of body-surface area every 3 weeks. The median overall survival was 9.2 months (95% confidence interval [CI], 7.3 to 13.3) with nivolumab versus 6.0 months (95% CI, 5.1 to 7.3) with docetaxel. The risk of death was 41% lower with nivolumab than with docetaxel (hazard ratio, 0.59; 95% CI, 0.44 to 0.79; P<0.001). At 1 year, the overall survival rate was 42% (95% CI, 34 to 50) with nivolumab versus 24% (95% CI, 17 to 31) with docetaxel. The response rate was 20% with nivolumab versus 9% with docetaxel (P=0.008). The median PFS was 3.5 months with nivolumab versus 2.8 months with docetaxel (hazard ratio for death or disease progression, 0.62; 95% CI, 0.47 to 0.81; P<0.001). The expression of the PD-1 ligand (PD-L1) was neither prognostic nor predictive of benefit. Treatment-related adverse events of Grade 3 or 4 were reported in 7% of the patients in the nivolumab group as compared with 55% of those in the docetaxel group (39).
The second trial, checkmate 057 used the same design in the non-squamous subgroup. Patients in the CheckMate 057 study had progressed after treatment with platinum-based doublet chemotherapy (and, if eligible, a tyrosine kinase inhibitor), a guideline-recommended first-line therapy for nonsquamous NSCLC. They were randomly assigned to subsequent treatment with nivolumab (3 mg/kg every 2 weeks; 292 patients) or docetaxel (75 mg/m2 every 3 weeks; 290 patients); both drugs were continued until progression or discontinuation due to toxicity (40).
The primary efficacy endpoint of the study was overall survival (OS). Treatment with nivolumab significantly improved median OS, with a hazard ratio for death of 0.73 (95% CI: 0.59, 0.89; P=0.00155) compared with docetaxel. One-year OS was 50.5% with nivolumab versus 39.0% with docetaxel. Other study endpoints included PFS, ORR, and nivolumab efficacy by PD-L1 expression.
Significantly more patients had an objective response (19.2% vs. 12.4%; P=0.0235). At the time of the analysis, the median duration of response to nivolumab was 17.1 months, compared with 5.6 months for docetaxel. No difference between nivolumab and docetaxel was observed in median PFS (2.3 months vs. 4.2 months; P=0.393). PD-L1 expression was associated with improved efficacy for patients treated with nivolumab, an effect most dramatically seen in patients with PD-L1 expression 5% or higher and 10% or higher, but evident at PD-L1 expression levels as low as 1% or higher.
Also of note, subgroup analysis favoured nivolumab over docetaxel in all categories, except patients 75 years of age or older, never smokers, and those positive for EGFR mutations. Treatment-related adverse reactions of grade 3 to 5 severity occurred at a higher rate with docetaxel (53.7%) than with nivolumab (10.5%).
Regarding toxicity, and as a summary, across all clinical trials performed to date using anti-PD‑1 drug monotherapy and in particular nivolumab, the observed incidence of severe pneumonitis is less than 5% with nivolumab monotherapy.
Chemotherapy stimulates an immune response against tumors, which may facilitate immunotherapy´s anticancer activity. Evidence of synergy between chemotherapy and immunotherapy was shown in several studies. The feasibility of combining both targeted agents and immunotherapy is also being studied in the CheckMate 012 trial and more recently in CheckMate 227 phase III study.
Neoadjuvant administration of two doses of nivolumab in patients with early stage lung cancer led to a major pathological response in 45% of tumors (41) . Major pathological response defined as <10% viable tumor cells in the resected specimen.
NADIM Study (CA209-547) (42) is a Phase II, single-arm, open-label multicenter study aimed to assess the feasibility, safety and efficacy of combined neoadjuvant chemotherapy and immunotherapy. (Figure 1) The primary endpoint is the progression-free survival (PFS) at 24 months from diagnosis .
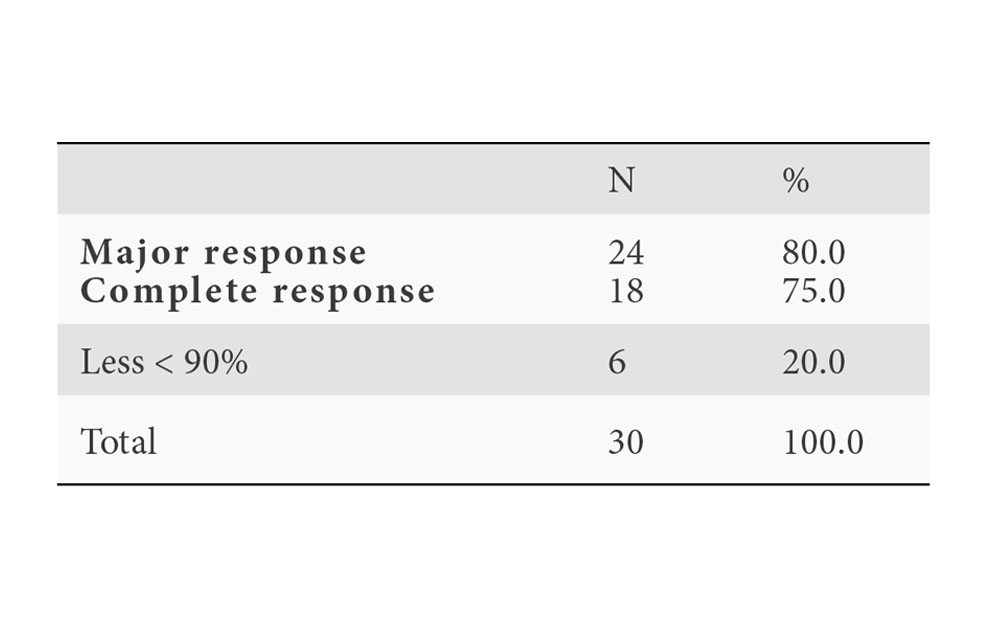
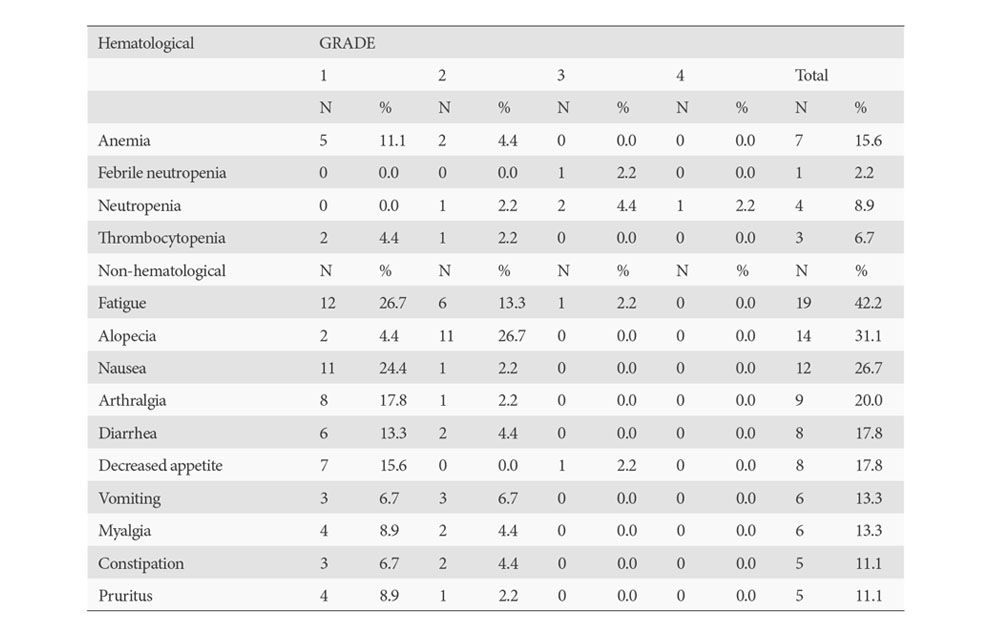
Results: (cut-off date 30th June 2018). Available efficacy results for this subset of 30 patients underwent surgery: No intraoperative complications were documented. 7/30 patients had postsurgical complications. There was no post-operative mortality. Clinical results: Tumor responses after neoadjuvant therapy (100% compliance rate), according to RECIST criteria v1.1 assessed per CT-SCAN: -ORR= 21/30 (70%), including 3 Complete Responses (CR) (10%) and 18 Partial Response (60%), Stable disease was reported for the remaining 9/30 (30%) patients. Pathological responses after surgery (Table 1): pCR rates observed are unprecedented and highly promising in the context of neoadjuvant therapy of NSCLC, with down-staging around 90% and without important toxicity (Table 2)
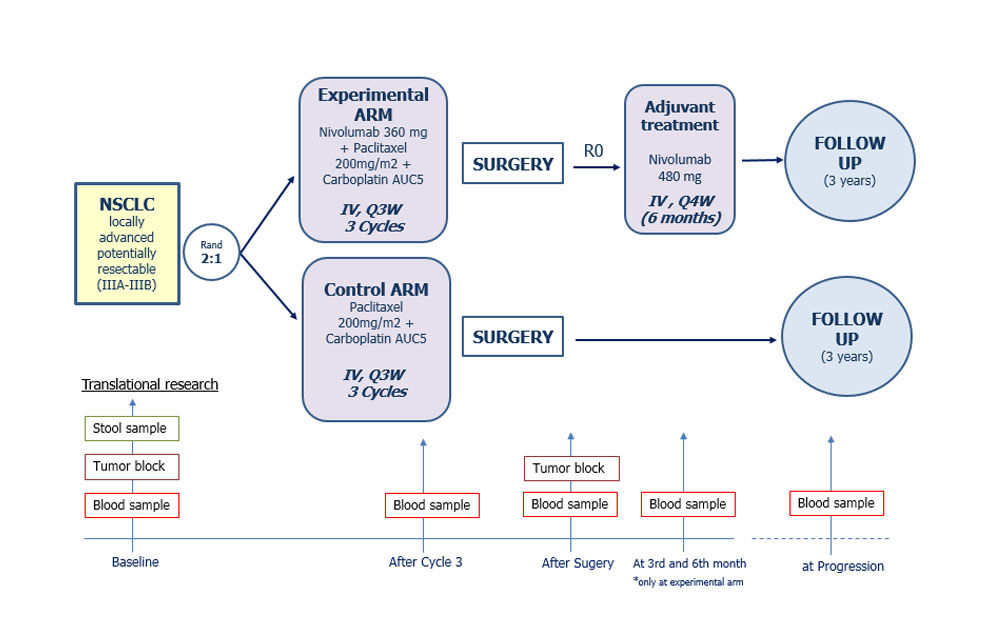
Current study results are considered an acceptable evidence to support the hypothesis of efficacy of the proposed Chemo Immunotherapy (Ch-IO) combination in the setting of neoadjuvant treatment in NSCLC but it is deemed not to reach the usual level of robustness needed to substantiate a new therapeutic indication due to the limitations previously outlined. To confirm these results a properly-designed and conducted randomized trial is mandatory to be able to address the limitations of the NADIM exploratory study and to generate a sound and sufficiently solid evidence of efficacy and safety and for that we will start NADIM II. (Figure 2. NADIM II)
We think that one of the most promising treatment modalities in stage III in lung cancer that has emerged in recent years is chemo-immunotherapy.
BIBLIOGRAFÍA
- Mountain CF. Revisions in the international system for staging lung cancer. Chest 1997; 111: 1710-1717.
- López-Abente G, Pollán M, Aragonés N et al. Situación del cáncer en España: incidencia. An Sist Sanit Navar 2004; 27 (2): 165-173.
- Ramalingam S, Belani C. Systemic chemotherapy for advanced non-small cell lung cancer: recent advances and future directions. Oncologist 2008; 1: 5-13.
- Hanna N, Neubauer M, Yiannoutsos et al. Phase III study of cisplatin, etoposide and concurrent chest radiation with or without consolidation with docetaxel in patients with inoperable stage III NSCLC: The Hoosier Oncology Group and U.S. Oncology. J Clin Oncol 2008; 26: 5755-5760.
- Douillard JY, Rosell R, De Lena M et al. Adjuvant vinorelbine plus cisplatin versus observation in patients with completely resected satge IB-IIIA NSCLC (Adjuvant Navelbine International Trialist Association (ANITA): a randomised controlled trial. Lancet Oncol 2006; 7: 719-727.
- Pignon JP, Tribodet H, Scagliotti GV et al. Lung adjuvant cisplatin evaluation: a pooled analysis by LACE Collaborative Group. J Clin Oncol 2008; 26: 3552-3559.
- Perez CA, Pajak TF, Rubin P et al. Long-term observations of the patterns of failure in patients with unresectable non oat cell carcinoma of the lung treated with definitive radiotherapy: report by the Radiation Therapy Oncology Group. Cancer 1987; 59: 1874–1881.
- Langer CJ, Leighton JC, Comis RL et al. Paclitaxel and carboplatin in combination in the treatment of advanced non-small-cell lung cancer (NSCLC): a phase II toxicity, response, and survival analysis (FCCC 93-024). J Clin Oncol 1995; 13: 1860-1870
- Non-Small Cell Lung Cancer Co-operative Group. Chemotherapy in non-small cell lung cancer: a meta-analysis using updated data on individual patients from 52 randomized clinical trials. Br Med J 1995; 311: 899-909.
- Martin J, Ginsberg RJ, Ennapadam S et al. Long-term results of combined-modality therapy in resectable non-small cell lung cancer. J Clin Oncol 2002; 20: 1989-1995.
- O´Brien MER, Splinter T, Smit EF et al. Carboplatin and paclitaxel (Taxol) as an induction regimen for patients with biopsy-proven stage IIIA N2 non-small cell lung cancer : an EORTC phase II study (EORTC 08958). Eur J Cancer 2003; 39: 1416-1422.
- Zaragoulidis K, Kontakiotis T, Hatziapostolou P et al. A phase II study of docetaxel and carboplatin in the treatment of non-small cell lung cancer. Lung Cancer 2001; 32: 281-287.
- Sugarbaker DJ, Herdon J, Kohman LJ et al. Results of cancer and leukemia group B protocol 8935: a multi-institutional phase II trimodality trial for stage IIIA (N2) non-small-cell lung cancer-Cancer and Leukemia Group B Thoracic Surgery Group. J Thorc Cardiovasc Surg 1995; 109: 473-485.
- Kirn DH, Lynch TJ, Mentzer SJ et al. Multimodality therapy of patients with satge IIIA, N2 non-small –cell lung cancer: impact of preoperative chemotherapy on resectability and downstaging,. J Thorac Cardiovasc Surg 1993; 106: 696-702.
- Martini N, Kris MG, Flehinger BJ et al. Preoperative chemotherapy for stage IIIa (N2) lung cancer: the Sloan-Kettering experience with 136 patients. Ann Thorc Surg 1993; 55: 1365-1374.
- Martín N. Mediatinal lymph node dissection for lung cancer: the memorial experience. Chest Surg Clin N Am 1995; 5: 189-203.
- Kumar P, Herdon J, Elias , et al. Comparison of pre-operative thoracic radiation therapy to pre-opetative chemotherapy on surgically staged IIIA (N2) non-small cell lung cancer: initial results of Cancer and Leucemia Group B Phase III Protocol 9134. Int J Radiat Oncol Biol Phys 1997; 39: 195.
- Choi Y S, shim Y M, Kim J, Kim K. Recurrence-free survival and prognostic factors in resected pN2 non-small cell lung cancer. Eur J Cardiothor Surg 2002; 22: 695-700.
- Miller AB, Hoogstraten B, Staquet M et al. Reporting results of cancer treatment. Cancer 1981; 47: 207-214.
- Martín N. Mediatinal lymph node dissection for lung cancer: The Memorial experience. Chest Surg Clin N Am 1995; 5: 189-203.
- Johnson DH, Paul DM, Hande KR et al. Paclitaxel plus carboplatin in advanced non-small-cell lung cancer – a phase II trial. J Clin Oncol 1996; 14: 2054-2060.
- Rowinsky EK, Flood WA, Sartorius SE, Bowling MK, Wagner J, Ettinger DS. Phase I study of paclitaxel as a 3-hour infusion followed by carboplatin in untreated patients with stage IV non-small-cell lung cancer. Semin Oncol 1995; 22 (Suppl 9): 48-54.
- Vafai D, Israel V, Zaretsky S, Natale RB. Phase III trial of combination carboplatin and taxol in non-small-cell lung cancer (NSCLC). Proc Am Soc Clin Oncol 1995; 14: A352.
- Klastersky J, Sculier JP, Lacroix H et al. A randomized study comparing cisplatin or carboplatin with etoposide in patients with advanced non-small-cell lung cancer:EORTC Protocol 07861. J Clin Oncol. 1990; 8(9): 1556-1562.
- Bonomi P, Finkelstein D, Ruckdeschel J et al. Combination Chemotherapy versus single agents followed by combination in stage IV non-small-cell lung cancer: a study of the ECOG. J Clin Oncol 1989; 7: 1602-1613.
- Greco FA y Hainsworth JD. Paclitaxel (1-hour infusion) plus carboplatin in the treatment of advanced NSCLC. Semin Oncol 1997; 24: S12-17.
- DeVore RF, Jagasia M y Johnson D. Paclitaxel by either 1-hour or 24h infusion in combination with carboplatin in advanced NSCLC. Semin Oncol 1997; 24: S27-29.
- Palackdharry CS, Durham SJ, Durzinsky DS et al: Preliminary results of neoadjuvant paclitaxel and carboplatin in the treatment of early stage NSCLC. Semin Oncol 1997; 24: S12-34-S12-36.
- Stroun M et al. Neoplastic characteristics of the DNA found in the plasma of cancer patients. Oncology 1989; 46:318-322.
- Decoster L, Wauters I, Vansteenkiste JF. Vaccination therapy for non-small-cell lung cancer: review of agents in phase III development. Ann Oncol 2012; 23: 1387-1393.
- Leach DR, Krummel MF, Allison JP. Enhancement of antitumor immunity by CTLA-4 blockade. Science 1996; 271: 1734-1736.
- Wing K, Sakaguchi S. Regulatory T cells exert checks and balances on self tolerance and autoimmunity. Nat Immunol 2010; 11: 7-13.
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 2012; 12: 252-264.
- Sharma P, Allison JP. The future of immune checkpoint therapy. Science 2015; 348: 56-61.
- Okazaki T, Maeda A, Nishimura H et al. PD-1 immunoreceptor inhibits B cell receptor-mediated signaling by recruiting src homology 2-domain-containing tyrosine phosphatase 2 to phosphotyrosine. Proc Natl Acad Sci USA 2001; 98: 13866-13871.
- Sheppard KA, Fitz LJ, Lee JM et al. PD-1 inhibits T-cell receptor induced phosphorylation of the ZAP70/CD3zeta signalosome and downstream signaling to PKCtheta. FEBS Lett 2004; 574: 37-41.
- Gettinger SN, Horn L, Gandhi L et al. Overall survival and long-term safety of nivolumab (Anti-Programmed Death 1 Antibody, BMS-936558, ONO-4538) in patients with previously treated advanced non-small-cell lung cancer. J Clin Oncol 2015; 33(18): 2004-2012.
- Topalian SL, Hodi FS, Brahmer JR et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med 2012; 366: 2443-2454.
- Brahmer J, Reckamp KL, Baas P et al. Nivolumab versus Docetaxel in Advanced Squamous-Cell Non-Small-Cell Lung Cancer. N Engl J Med 2015; 373: 123-135.
- Paz-Ares L, Horn L, Borghaei H et al. Phase III, randomized trial (CheckMate 057) of nivolumab (NIVO) versus docetaxel (DOC) in advanced non-squamous cell (non-SQ) non-small cell lung cancer (NSCLC). J Clin Oncol 2015; 33, (suppl; abstr LBA109).
- Forde PM, Chaft JE, Smith V, et al. Neoadjuvant PD-1 blockade in resectable lung cancer. N Engl J Med 2018. 24; 378(21):1976-1986.
- Provencio M, Nadal E et al. Neo-adjuvant chemo/immunotherapy for resectable stages IIIA non-small cell lung cancer: Nadim Study-SLCG IASLC World Lung Cancer Congress. Toronto; 2018.
DECLARACIÓN DE TRANSPARENCIA
El autor/a de este artículo declara no tener ningún tipo de conflicto de intereses respecto a lo expuesto en la presente revisión.
ranm tv
Mariano Provencio Pulla
Real Academia Nacional de Medicina de España
C/ Arrieta, 12 · 28013 Madrid
Tlf.: +34 91 159 47 34 | mprovenciop@gmail.com
Año 2019 · número 136 (01) · páginas 17 a 24
Enviado*: 26.02.19
Revisado: 04.03.19
Aceptado: 28.03.19
* Fecha de lectura en la RANM


